Chapter 3: Landfill Gas Safety and Health Issues
This chapter provides information about health and safety issues associated with landfill gas—specifically, possible explosion and asphyxiation hazards and issues related to odors emanating from the landfill and low-level chemical emissions. It also contains information about health and safety issues associated with landfill fires (which may or may not be the direct result of landfill gas). This chapter also describes the tools that can be used to help environmental professionals respond to community health concerns. It provides information about what is known and unknown about the short-term and long-term health effects associated with landfill gas emissions, which can be mixtures of hundreds of different gases.
When reading this chapter, keep in mind that if people are not being exposed to landfill gases, no adverse health effects are expected. Exposures occur only if the landfill is producing harmful levels of gases and if the gases are migrating from the landfills and reaching people. Responding to community concerns about the possible health impacts of known or potential landfill gas emissions can often be difficult. Data (at the point of exposure) are needed to fully evaluate exposures, and these data are often limited or not available (see Chapter Four).How are people exposed to landfill gas?
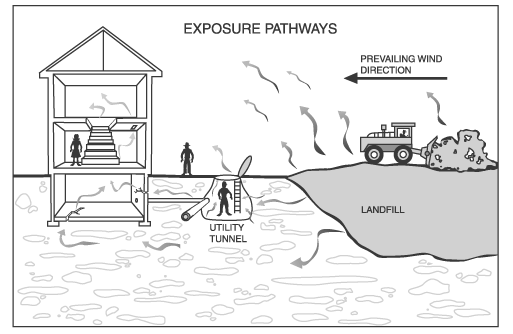
People may be exposed to landfill gases either at the landfill or in their communities. As discussed in Chapter Two, landfill gases may migrate from the landfill either above or below ground. Gases can move through the landfill surface to the ambient air. Once in the air, the landfill gases can be carried to the community with the wind. Odors from day-to-day landfill activities are indicative of gases moving above ground. Gases may also move through the soil underground and enter homes or utility corridors on or adjacent to the landfill. Figure 3-1 illustrates the movement of landfill gases and potential exposure pathways. The levels of gases that migrate from a landfill and to which people are exposed are dependent on many factors, as described in Chapter Two. Landfill gas collection and control systems have the greatest impact on gas migration and exposures. If a collection or control system is in place and operating properly, migration and exposures should be minimal.
Explosion Hazards
Landfill gas may form an explosive mixture when it combines with air in certain proportions. This section provides information about:
- The conditions that must be met for landfill gas to pose
an explosion hazard.
- The types of gases that may potentially pose explosion hazards.
- What can be done to assess whether a landfill is posing
an explosion hazard.
When does landfill gas pose an explosion hazard?
The following conditions must be met for landfill gas to pose an explosion hazard:
- Gas production. A landfill must be producing
gas, and this gas must contain chemicals that are present at
explosive levels.
- Gas migration. The gas must be able to
migrate from the landfill. Underground pipes or natural subsurface
geology may provide migration pathways for landfill gas (see
Chapter Two, "What factors affect landfill gas migration?").
Gas collection and treatment systems, if operating properly,
reduce the amount of gas that is able to escape from the landfill.
(See
Chapter Five.)
- Gas collection in a confined space. The gas must collect in a confined space to a concentration at which it could potentially explode. A confined space might be a manhole, a subsurface space, a utility room in a home, or a basement. The concentration at which a gas has the potential to explode is defined in terms of its lower and upper explosive limits (LEL and UEL), as defined at right.
Lower and Upper Explosive Limits (LEL and UEL)
The concentration level at which gas has the potential to explode is called the explosive limit. The potential for a gas to explode is determined by its lower explosive limit (LEL) and upper explosive limit (UEL). The LEL and UEL are measures of the percent of a gas in the air by volume. At concentrations below its LEL and above its UEL, a gas is not explosive. However, an explosion hazard may exist if a gas is present in the air between the LEL and UEL and an ignition source is present.
|
Landfill Gas Explosions
|
|
|
Although landfill gas explosions are by no means common occurrences, a number of incidents known or suspected to have been caused by landfill gas explosions have been documented. |
|
| 1999 | An 8-year-old girl was burned on her arms and legs when playing in an Atlanta playground. The area was reportedly used as an illegal dumping ground many years ago. (Atlanta Journal-Constitution 1999) |
| 1994 | While playing soccer in a park built over an old landfill in Charlotte, North Carolina, a woman was seriously burned by a methane explosion. (Charlotte Observer 1994) |
| 1987 | Off-site gas migration is suspected to have caused a house to explode in Pittsburgh, Pennsylvania.(EPA 1991) |
| 1984 | Landfill gas migrated to and destroyed one house near a landfill in Akron, Ohio.Ten houses were temporarily evacuated. (EPA 1991) |
| 1983 | An explosion destroyed a residence across the street from a landfill in Cincinnati, Ohio. Minor injuries were reported. (EPA 1991) |
| 1975 | In Sheridan, Colorado, landfill gas accumulated in a storm drain pipe that ran through a landfill. An explosion occurred when several children playing in the pipe lit a candle, resulting in serious injury to all the children. (USACE 1984) |
| 1969 | Methane gas migrated from an adjacent landfill into the basement of an armory in Winston-Salem, North Carolina. A lit cigarette caused the gas to explode, killing three men and seriously injuring five others. (USACE 1984) |
See the box above for a few of many documented situations where all the conditions for explosions were met and explosions actually occurred.
What types of gases can pose an explosion hazard?
- Methane. Methane is the constituent of
landfill gas that is likely to pose the greatest explosion hazard.
Methane is explosive between its LEL of 5% by volume and its
UEL of 15% by volume. Because methane concentrations within
the landfill are typically 50% (much higher than its UEL), methane
is unlikely to explode within the landfill boundaries. As methane
migrates and is diluted, however, the methane gas mixture may
be at explosive levels. Also, oxygen is a key component for
creating an explosion, but the biological processes that produce
methane require an anaerobic, or oxygen-depleted, environment.
At the surface of the landfill, enough oxygen is present to
support an explosion, but the methane gas usually diffuses into
the ambient air to concentrations below the 5% LEL. In order
to pose an explosion hazard, methane must migrate from the landfill
and be present between its LEL and UEL.
- Other landfill gases. Other landfill gas constituents (e.g., ammonia, hydrogen sulfide, and NMOCs) are flammable. However, because they are unlikely to be present at concentrations above their LELs, they rarely pose explosion hazards as individual gases. For example, benzene (an NMOC that may be found in landfill gas) is explosive between its LEL of 1.2% and its UEL of 7.8%. However, benzene concentrations in landfill gas are very unlikely to reach these levels. If benzene were detected in landfill gas at a concentration of 2 ppb (or 0.0000002% of the air by volume), then benzene would have to collect in a closed space at a concentration 6 million times greater than the concentration found in the landfill gas to cause an explosion hazard.
Table 3-1 summarizes the potential explosion hazards posed by the important constituents of landfill gas. Keep in mind that methane is the most likely landfill gas constituent to pose an explosion hazard. Other flammable landfill gas constituents are unlikely to be present at concentrations high enough to pose an explosion hazard. However, the flammable NMOCs do contribute to total explosive hazard when combined with methane in a confined space.
How can I assess whether a landfill in my community poses an explosion hazard?
The checklist on the following page can help determine if a landfill may pose an explosion hazard. If your evaluation identifies the potential for an explosion, several actions can be taken to prevent harm to the community. Measures and controls to prevent explosion hazards are discussed in Chapter Five. Possible public health actions are described in Appendix B.
Asphyxiation Hazards
Landfill gas poses an asphyxiation hazard only if it collects in an enclosed space (e.g., a basement or utility corridor) at concentrations high enough to displace existing air and create an oxygen-deficient environment. The Occupational Safety and Health Administration (OSHA) defines an oxygen-deficient environment as one that has less than 19.5% oxygen by volume (OSHA n.d.a). Ambient air contains approximately 21% oxygen by volume. Health effects associated with oxygen-deficient environments are described in Table 3-2.
Any of the gases that comprise landfill gas can, either individually or in combination, create an asphyxiation hazard if they are present at levels sufficient to create an oxygen-deficient environment.
Carbon dioxide, which comprises 40% to 60% of landfill gas, may pose specific asphyxiation hazard concerns. Because it is denser than air, carbon dioxide that has escaped from a landfill and collected in a confined space, such as a basement or an underground utility corridor, may remain in the area for hours or days after the area has been opened to the air (e.g., after a manhole cover has been removed or a basement door opened). Carbon dioxide is colorless and odor-less and therefore not readily detectable. Carbon dioxide concentrations of 10% or more can cause unconsciousness or death. Lower concentrations may cause headache, sweating, rapid breathing, increased heartbeat, shortness of breath, dizziness, mental depression, visual disturbances, or shaking. The seriousness of these symptoms depends on the concentration and duration of exposure. The response to carbon dioxide inhalation varies greatly even in healthy normal individuals.
In assessing the public health issues of migrating landfill gas,
environmental health professionals should investigate the presence
of buried utility lines and storm sewers on or adjacent to the landfill.
These structures not only provide a pathway for migrating gases,
but also pose a special asphyxiation problem for utility workers
who fail to follow confined space entry procedures prescribed by
OSHA. On-site or adjacent residences and commercial buildings with
basements or insulated (or sealed) crawl spaces should also be investigated
for potential asphyxiation hazards.
Health Issues Associated with Landfill Gas Emissions - Odors and Low-Level Chemical Exposures
Landfill odors often prompt complaints from community members. People may also have concerns about health effects associated with these odors and other emissions coming from the landfill. This section contains information about
- Symptoms possibly triggered by landfill gas odors.
- What scientists know about the potential health effects
of exposures to landfill gas emissions.
- How environmental health professionals can assess whether landfill gas emissions may be posing a health threat.
Can the presence of odors trigger symptoms?
People in communities near landfills are often concerned about odors emitted from landfills. They say that these odors are a source of undesirable health effects or symptoms, such as headaches and nausea. At low-level concentrations—typically associated with landfill gas— it is unclear whether it is the constituent itself or its odors that trigger a response. Typically, these effects fade when the odor can no longer be detected. The box below describes the biology behind detecting odors.
Landfill gas odors are produced by bacterial or chemical processes and can emanate from both active or closed landfills. These odors can migrate to the surrounding community. Potential sources of landfill odors include sulfides, ammonia, and certain NMOCs, if present at concentrations that are high enough. Landfill odors may also be produced by the disposal of certain types of wastes, such as manures and fermented grains.
| How Do People Detect Odors? |
|
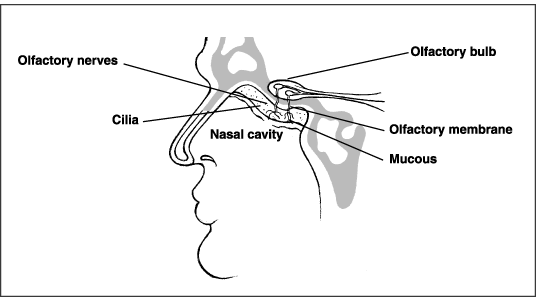
Humans can distinguish between 3,000 and 10,000 different odors. Although people commonly believe that they smell with their noses, the nose is only one part of the olfactory system that allows humans to distinguish smells. The nose serves as a vacuum that pulls in air and odorous chemicals, such as hydrogen sulfide. The air and odorous chemicals are warmed in the nasal cavity and then trapped in mucus surrounding the olfactory membrane. The olfactory membrane is an area smaller than 1 square inch located deep in the nasal cavity between the eyes. Odorous chemicals interact with receptors (chemoreceptors) found on small hairs (cilia) on the olfactory membrane. The receptors send messages about the odorous chemicals to the brain through the olfactory bulb. The brain then interprets and identifies the odor (Pacific Science Center 1999; Jacobs 1999). The sense of smell, just like the other senses of sight, hearing, taste, and touch, varies from person to person. One person may be able to smell an odor like hydrogen sulfide at extremely low concentrations, while another person in the same community or home cannot. Because of this variation, there is no true odor threshold value above which odors are unpleasant and below which odors are not noticeable. Any odor threshold values reported in the literature, such as those provided in Table 3-3, are only estimates of concentrations that the average person may detect (AIHA 1989). Therefore, health professionals should be cautious when citing established threshold values or discussing them with community members. Anatomy of Smell |
- Sulfides. Hydrogen sulfide, dimethyl sulfide,
and mercaptans are the three most common sulfides responsible
for landfill odors. These gases produce a very strong rotten-egg
smell—even at very low concentrations. Of these three sulfides,
hydrogen sulfide is emitted from landfills at the highest rates
and concentrations.
Humans are extremely sensitive to hydrogen sulfide odors and can smell such odors at concentrations as low as 0.5 to 1 part per billion (ppb). At levels approaching 50 ppb, people can find the odor offensive. Average concentrations in ambient air range from 0.11 to 0.33 ppb (ATSDR 1999a). According to information collected by the Connecticut Department of Health, the concentration of hydrogen sulfide in ambient air around a landfill is usually close to 15 ppb (CTDPH 1997; ATSDR 1999a).
- Ammonia. Ammonia is another odorous landfill
gas that is produced by the decomposition of organic matter
in the landfill. Ammonia is common in the environment and an
important compound for maintaining plant and animal life. People
are exposed daily to low levels of ammonia in the environment
from the natural breakdown of manure and dead plants and animals.
Because ammonia is commonly used as a household cleaner, most
people are familiar with its distinct smell.
Humans are much less sensitive to the odor of ammonia than they are to sulfide odors. The odor threshold for ammonia is between 28,000 and 50,000 ppb. Landfill gas has been reported to contain between 1,000,000 and 10,000,000 ppb of ammonia, or 0.1% to 1% ammonia by volume (Zero Waste America n.d.). Concentrations in ambient air at or near the landfill site are expected to be much lower.
- NMOCs. Some NMOCs, such as vinyl chloride and hydrocarbons, may also cause odors. In general, however, NMOCs are emitted at very low (trace) concentrations and are unlikely to pose a severe odor problem.
Table 3-3 lists some of the common landfill gas components and their odor thresholds.
Many people may find the odors emitted from a landfill offensive or unpleasant. In reaction to the odor, some people may experience nausea or headaches. Although such responses are undesirable, medical attention is usually not required. Often, symptoms such as headaches and nausea fade when the odor goes away. However, the effects on day-to-day life can be more lasting. Families living close to a landfill in Connecticut described frequent odor events as overwhelmingly disruptive. One family reported being awakened during predawn hours by a flood of nauseating air that persisted for 2 or more hours. The loss of sleep and the frustration from the frequent odor events greatly added to the level of stress in the family's life. Although landfill odors may not associated with long-term adverse health effects or illness for most people, the added disruption and stress of day-to-day activities can greatly impact quality of life. The story below describes how environmental and health professionals addressed odor problems in Danbury, Connecticut.
| The Danbury Landfill—One Community's Story |
|
Danbury, Connecticut, is a community that successfully tackled a landfill odor problem. In the Spring of 1996, a 100-year-old landfill in Danbury caught fire. Water used to extinguish the fire promoted bacteria growth and increased the production of odor-causing sulfides, especially hydrogen sulfide. The increase in odors prompted public concerns and questions. Though hydrogen sulfide levels in the air were well below concentrations that might affect human health, the odor caused nausea and headaches in some residents. Local and state health authorities and environmental agencies worked together to address the odor problem. They took the following actions to alleviate community concerns and address the odor problem:
The first action level was triggered a few times during the period when odor control measures were being installed. The other action levels were never triggered. Once odors were controlled, community complaints decreased markedly. |
The impact of landfill gas odors on sensitive populations such as people with pre-existing respiratory illnesses is not well documented or understood. A study conducted on Staten Island showed an increase in self-reported wheezing among asthmatics living near a landfill on days when they reported odors. Ambient air measurements, however, showed levels of hydrogen sulfide and other emissions much lower than levels known to be associated with adverse health effects (ATSDR 1999b). The box below provides more information about this study. The study suggests that odors in and of themselves may trigger respiratory effects among asthmatics. This preliminary conclusion may be confounded by other environmental triggers for respiratory response in asthmatics, such as dust mites, animal dander, tobacco smoke, and outdoor air pollution. The American Lung Association Web site ( http://www.lungusa.org/lung-disease/asthma/) provides more information about possible environmental triggers for asthma. EPA provides information about asthma itself at http://www.epa.gov/asthma/index.html.