What Respiratory Conditions Are Associated with Asbestos?
According to the American Thoracic Society [2004], “asbestos has been the largest cause of occupational cancer in the United States and a significant cause of disease and disability from nonmalignant disease.” It has been estimated that the cumulative total number of asbestos-associated deaths in the United States may exceed 200,000 by the year 2030 [Nicholson et al. 1982].
Depending on the level and duration of exposure, inhalation of asbestos fibers can cause different diseases such as
- Asbestosis,
- Asbestos-related pleural abnormalities,
- Lung and laryngeal carcinoma, and/or
- Malignant mesothelioma of the pleura or peritoneum.
Any combination of these diseases can be present in a single patient. Clinically, it is important to distinguish nonmalignant conditions from malignant diseases; differential diagnosis will be discussed further in later sections of this document.
Asbestosis is a diffuse interstitial fibrosis of lung tissue resulting from inhalation of asbestos fibers. Asbestos fibers inhaled deep into the lung become lodged in the tissue, eventually resulting in diffuse alveolar and interstitial fibrosis. The fibrosis usually first occurs in the respiratory bronchioles, particularly in the subpleural portions of the lower lobes. The fibrosis can progress to include the alveolar walls. Fibrosis tends to progress even after exposure ceases [Khan et al. 2013]. This fibrosis can lead to
- Reduced lung volumes,
- Decreased lung compliance,
- Impaired gas exchange,
- Restrictive pattern of impairment,
- Obstructive features due to small airways disease, and
- Progressive exertional dyspnea with an insidious onset.
Asbestosis is characterized by the following radiographic changes: fine, irregular opacities in both lung fields (especially in the bases) and septal lines that progress to honeycombing and sometimes, in more severe disease, obscuration of the heart border and hemi-diaphragm – the so-called shaggy heart sign [Khan et al. 2013]. Radiographic changes depend on the
- Duration,
- Frequency, and
- Intensity of exposure.
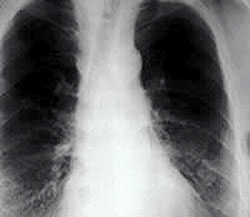
Figure 5. Chest radiograph of asbestosis in the lung
Patients with asbestosis may have elevated levels of antinuclear antibody and rheumatoid factors and a progressive decrease in total lymphocyte count with advancing fibrosis.
Asbestosis has no unique pathognomonic signs or symptoms, but diagnosis is made by the constellation of clinical, functional, and radiographic findings as outlined by the American Thoracic Society [American Thoracic Society 2004]. These criteria include
- Sufficient history of exposure to asbestos,
- Appearance of disease with a consistent time interval from first exposure,
- Clinical picture such as insidious onset of dyspnea on exertion, bibasilar end-inspiratory crackles not cleared by coughing,
- Functional tests showing restrictive (occasionally obstructive) pattern with reduced diffusing capacity (DLco),
- Characteristic radiographic appearance, and
- Exclusion of other causes of interstitial fibrosis or obstructive disease such as usual interstitial pneumonia, connective tissue disease, drug-related fibrosis [American Thoracic Society 2004; Khan et al. 2013].
The table below describes the natural history associated with asbestosis.
| Parameter | Typical Findings |
|---|---|
| Sufficient exposures | Usually associated with high-level occupational exposures [Khan et al. 2013]. |
| Latency periods | Radiographic changes: commonly <20 years. Clinical manifestations: commonly 20-40 years. Asbestosis appears earliest in those with the highest exposure levels [American Thoracic Society 2004]. |
| Risk of asbestosis | Asbestosis develops in around 50% of adults with occupational asbestos exposure [Khan et al. 2013]. |
| Co-morbid conditions | Increased risk for asbestos-related lung cancer and mesothelioma, though both can occur without asbestosis [Weiss 1999; Hillerdal 1999]. |
| Mortality and Morbidity | Severe asbestosis may lead to respiratory failure over 1-2 decades. Many patients with asbestosis die of other causes such as asbestos-associated lung cancer (38%), mesothelioma (9%), and other causes (32%) [Rosenberg 1997, Kamp and Weitzman 1997]. |
Asbestos-related pleural abnormalities encompass four types of pleural changes.
- Pleural plaques.
- Non-malignant asbestos pleural effusions.
- Diffuse pleural thickening.
- Rounded atelectasis (folded lung).
The pleura are more sensitive to asbestos than the lung parenchyma, so the effects of asbestos exposure show here first and occur at much lower doses than the fibrotic changes in the lung [Peacock et al. 2000; Khan et al. 2013].
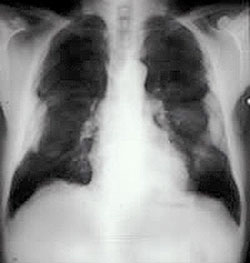
Figure 6. Chest radiograph showing bilateral pleural plaques
- Ceramic fibers,
- Talc,
- Silicates,
- Erionite, a rare zeolite [Finkelstein 2013; Clark et al. 2006; Zenden et al. 1997].
Non-malignant asbestos pleural effusions are small and often bloody unilateral effusions [Khan et al. 2013]. These effusions are among the earliest manifestations of asbestos exposure; they can occur within 10 years of exposure [Chapman et al. 2003]. They are usually asymptomatic. Rarely, they can cause pain, fever, and dyspnea. These effusions typically last for months, and may occasionally recur. Their presence can precede the occurrence of diffuse pleural thickening [Chapman et al. 2003].
Diffuse pleural thickening is a non-circumscribed fibrous thickening of the visceral pleura with areas of adherence to the parietal pleura and obliteration of the pleural space. It can be associated with more extensive asbestos exposure than pleural plaques [Chapman et al. 2003]. In fact, diffuse pleural thickening has been reported in 10% of patients with asbestosis [Khan et al. 2013]. Diffuse pleural thickening can occur after non-malignant pleural effusions. The fibrotic areas are ill-defined, involving costophrenic angles, apices, lung bases, and interlobar fissures. Diffuse pleural thickening can be associated with mild (or, rarely, moderate to severe) restrictive pulmonary function deficits such as decreased ventilatory capacity. When this occurs, the patient may experience progressive dyspnea and chest pain [Chapman et al. 2003].
Rounded atelectasis (or folded lung) occurs when portions of lung tissue are caught in bands of fibrous pleural tissue with in-drawing of the bronchi and vessels [Khan et al. 2013]. This produces a distinctive radiographic appearance: a rounded pleural mass with bands of lung tissue radiating outwards. This condition is usually asymptomatic, though some patients develop dyspnea or dry cough. The course is usually stable or slowly progressive. Folded lung is the least common asbestos-related non-malignant pleural disease; it is not only associated with asbestosis exposure but can occur following other exposures and medical conditions. However, asbestos exposure is the leading cause of rounded atelectasis, accounting for 29%-86% of cases. It can rarely also co-occur with lung cancer [Stathopoulos et al. 2005].
The differential causes of rounded atelectasis includes
- Exposure to mineral dusts such as asbestos, and occupational exposures to silica and mixed mineral dusts;
- Exudative pleural effusions such as
- Empyema,
- Tuberculous effusions,
- Hemothorax,
- Post-cardiac surgery,
- Chronic hemodialysis; and
- Other medical conditions such as
- Legionella pneumonia,
- Histoplasmosis,
- End-stage renal disease,
- Pneumothorax, and
- Childhood cancer [Stathopoulos et al. 2005].
The table below shows typical findings and natural history associated with asbestos-related non-malignant pleural abnormalities.
| Parameter | Pleural Plaques | Pleural Effusions | Diffuse Pleural Thickening | Rounded Atelectasis |
|---|---|---|---|---|
| Typical Exposures | Can occur with short low-level exposures or high-level occupational exposures. | Usually associated with moderate- to high-level exposures. Less specific for asbestos exposure than pleural plaques | May occur with occupational and environmental exposures. Has other causes besides asbestos exposure. | |
| Average Latency Periods | 20-30 years | 10 years | 15 years | N/A |
| Co-morbid Conditions | Since the presence of these plaques is an indicator of asbestos exposure, there is an increased incidence of asbestos-related diseases associated with them. | Other asbestos-related diseases. | Other asbestos-related diseases. Can follow non-malignant pleural effusions. | Follows non-malignant pleural effusions; can co-exist with other asbestos-related diseases or its other causes. |
| Mortality and Morbidity | Not fatal/usually asymptomatic; incidental finding. | Not fatal. Clinical presentation ranges from asymptomatic to pleuritic chest pain and fever. |
Not fatal. If severe, can cause dyspnea. Usually no significant functional impairment unless very extensive. |
Not fatal. Usually asymptomatic; if severe, chest pain, dyspnea, and cough. Usually no functional impairment unless accompanied by other asbestos-related disease. |
Most lung cancers are associated with exposure to tobacco smoke. According to the 2004 Surgeon General Report on smoking, men with a lifetime of smoking have a 16 percent higher risk of dying from lung cancer than those who do not smoke [Surgeon General 2004] Lung cancer currently accounts for 28% of all cancer deaths in the United States [Minna 2005].
Exposure to asbestos is associated with all major histological types of lung carcinoma (adenocarcinoma, squamous cell carcinoma, and small-cell carcinoma). It is estimated that 4%-12% of lung cancers are related to occupational levels of exposure to asbestos [Henderson et al. 2004. It is estimated that 20%-25% of heavily exposed asbestos workers will develop bronchogenic carcinomas [Khan et al. 2013]. Whether asbestos exposure will lead to lung cancer depends on several factors:
- Level, duration, and frequency of asbestos exposure (cumulative exposure) [Henderson et al. 2004],
- Time elapsed since exposure occurred,
- Age when exposure occurred,
- History of tobacco use, and
- Individual susceptibility factors not yet determined.
Most asbestos-related lung cancers reflect the dual influence of asbestos exposure and smoking [Henderson et al. 2004]. It has been known for over 25 years, that smoking and asbestos exposure have a multiplicative effect on the risk of lung cancer [Lee 2001; Henderson et al. 2004; ATSDR 2001]. The presence of asbestosis is an indicator of high-level asbestos exposure, but lung cancer can occur without asbestosis.
One of the best known sets of criteria to guide the clinician regarding whether asbestos contributed to lung cancer in an asbestos-exposed individual is the Helsinki criteria. For these criteria, some of the markers for attributing asbestos exposure as a contributing factor to lung cancer are
- The presence of asbestosis – serves as marker for significant exposures to asbestos,
- Estimated cumulative exposure to asbestos of at least 25 fiber-years (if known),
- By history, at least 1 year of heavy occupational exposure or 5-10 years of moderate exposure, and
- Lag time of at least 10 years since first exposure [Henderson et al. 1997].
The table below shows typical findings associated with lung carcinoma.
| Parameter | Typical Findings |
|---|---|
| Typical exposures | Large cumulative exposure (short-term, high-level exposures or long-term, moderate-level exposures). |
| Latency periods | 20-30 years. |
| Clinical presentation | Only 5%-15% of patients are asymptomatic when diagnosed. Most present with cough, hemoptysis, wheeze, dyspnea [Minna 2005]. |
| Co-morbid conditions | Asbestosis, other asbestos-related diseases. Paraneoplastic syndromes associated with lung cancer. |
| Mortality | Same as lung carcinoma with other causes – 14% five-year survival rate [British Thoracic Society 2001]. |
Diffuse malignant mesothelioma is a tumor arising from the thin serosal membrane of the body cavities:
- Pleura,
- Peritoneum,
- Pericardium,
- Tunica vaginalis testis, and
- Outer surface of ovaries.
It is a rare neoplasm, accounting for less than 5% of pleural malignancies. There are three histological types of malignant mesothelioma:
- Epithelial,
- Mixed, and
- Sarcomatous.
Of malignant mesotheliomas, 80% affect the pleura, and 20% affect the peritoneum [Khan et al. 2013]. A 2003 report of mesothelioma incidence in Australia reported pleural mesotheliomas at 93.2%, peritoneal mesotheliomas at 6.5% and mesotheliomas of other rare sites at 0.3% [Leigh and Driscoll 2003]. Peritoneal mesothelioma is discussed in the next section.
In most cases of pleural mesothelioma, the tumor is rapidly invasive locally [Lee et al. 2000]. Patients with malignant pleural mesothelioma can have sudden onset of pleural effusion and/or pleural thickening, dyspnea, and chest pain. By the time symptoms appear, the disease is most often rapidly fatal [British Thoracic Society 2001].
Pleural mesothelioma is a signal tumor for asbestos exposure; other causes are uncommon. The risk of mesothelioma depends on the amount of asbestos exposure [Weill et al. 2004]. All types of asbestos can cause mesothelioma, but some researchers believe that the amphibole form is more likely to induce mesothelioma than the serpentine form [ATSDR 2001a].
In 2007, about 2,700 people in the United States died of mesothelioma [NIOSH 2012a]. According to the National Cancer Institute’s Surveillance Epidemiology and End Results [SEER] data, there was an increase in the incidence of mesothelioma in the United States from the early 1970s to the mid-1990s, as disease developed in people exposed during peak asbestos exposure years (1940-1970). Mesothelioma incidence has probably started to decline in the United States, although it may still be increasing in Europe and Australia because of more abundant and prolonged use of asbestos in these countries than in the United States [Weill et al. 2004].
The table below shows typical findings associated with pleural mesothelioma.
| Parameter | Typical Findings |
|---|---|
| Typical exposures | Short-term, high-level exposures or chronic low-level exposures, especially to amphibole asbestos; incidence increases in dose-related manner [Hillerdal 1999]. |
| Latency periods | 10-57 years (30-40 years typical). |
| Clinical presentation | Frequently presents with chest pain accompanied by pleural mass or pleural effusion on chest radiograph [British Thoracic Society 2001]. |
| Mortality | High. The typical 1-year survival rate is <30%. Median survival time is 8-14 months after diagnosis [British Thoracic Society 2001]. |
- Asbestosis is an interstitial pulmonary fibrosis resulting from inhalation of asbestos fibers. It produces a restrictive pattern of disease and progressive exertional dyspnea.
- Asbestos-related pleural abnormalities include
- Pleural plaques,
- Non-malignant pleural effusions,
- Diffuse pleural thickening, and
- Rounded atelectasis.
They are relatively benign conditions, though diffuse pleural thickening can cause restrictive deficits.
- Lung carcinoma caused by asbestos exposure is histologically similar to lung cancer from other causes.
- Malignant pleural mesothelioma is a rare tumor arising from the pleural mesothelium. It is a signal tumor for asbestos exposure.
- Asbestos-associated respiratory diseases have long latency periods: 10 to 40 years or more, depending on the disease and exposure factors.