Pease Study

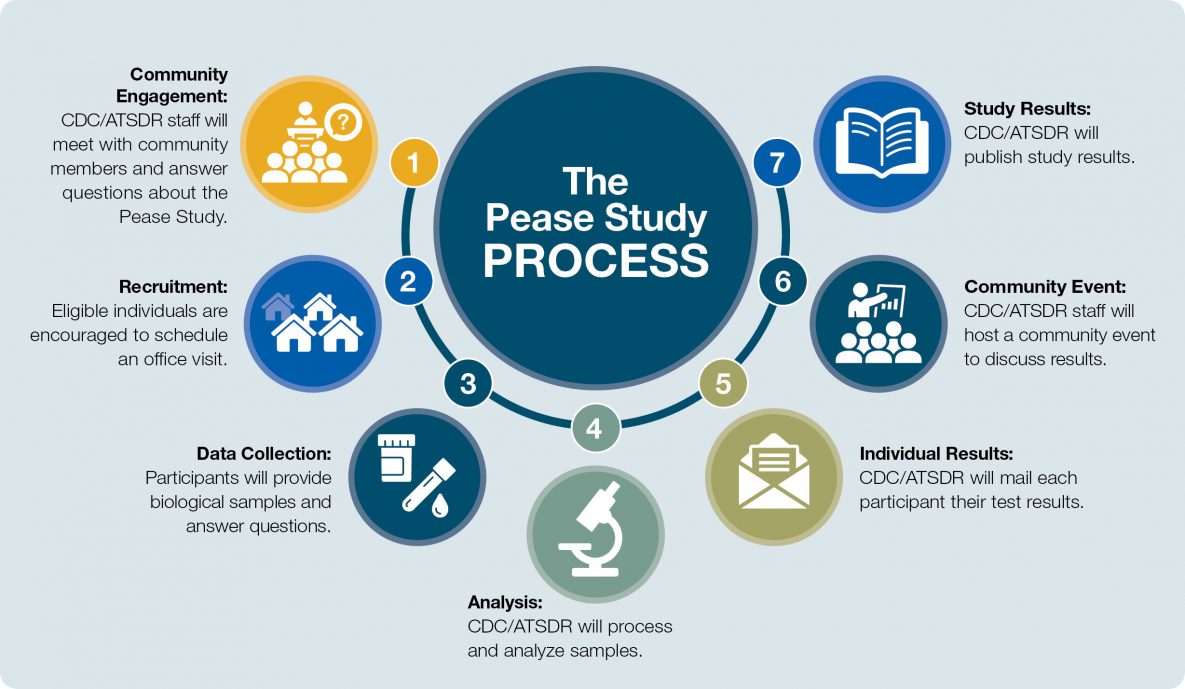
The Pease study report [PDF – 2 MB] is the first site of the national, Multi-site Study, which looks at the human health effects of Per- and Polyfluoroalkyl Substances (PFAS) exposure through drinking contaminated water. CDC/ATSDR will use the Pease Study to evaluate and improve procedures and methods for the Multi-site Study design.
As of December 31, 2021, Pease Study enrollment has ended. CDC/ATSDR is processing and analyzing blood and urine samples and will mail individual results to participants. CDC/ATSDR will write a final report summarizing the findings and recommendations and will share this information with the community.
Eligibility for the Pease Study included:
- Adults aged 18 or older who worked or attended school at Pease International Tradeport at any time from January 2004 to May 2014, or
- Who lived in Newington, NH, at any time from January 2004 to the present and used a private well with documented PFAS contamination.
- Children aged 4 through 17 who received parent or guardian study participation permission and who attended daycare at Pease International Tradeport at any time from 2004 to May 2014 or
- Were born to and/or breastfed by a mother who met the adult eligibility criteria were eligible.
- A small group of adults who never drank water from Pease International Tradeport and whose occupation or education did not expose them to PFAS, and children who never drank water from Pease International Tradeport, were also eligible to participate.
Individuals who had ever been employed as a firefighter, participated in fire training exercises using aqueous film-forming foam (AFFF), or employed at industrial facilities that used PFAS chemicals in the manufacturing process were not eligible to participate in the study. More specifically, it would be difficult to determine if elevated PFAS levels were from drinking contaminated water or from their exposure to PFAS at work. The Pease Study is learning more about exposure to contaminated drinking water. Similarly, children born to mothers who had any of these occupational exposures were not eligible to participate in the study either.
If you have questions about the study, please email us at PeaseStudy@cdc.gov.
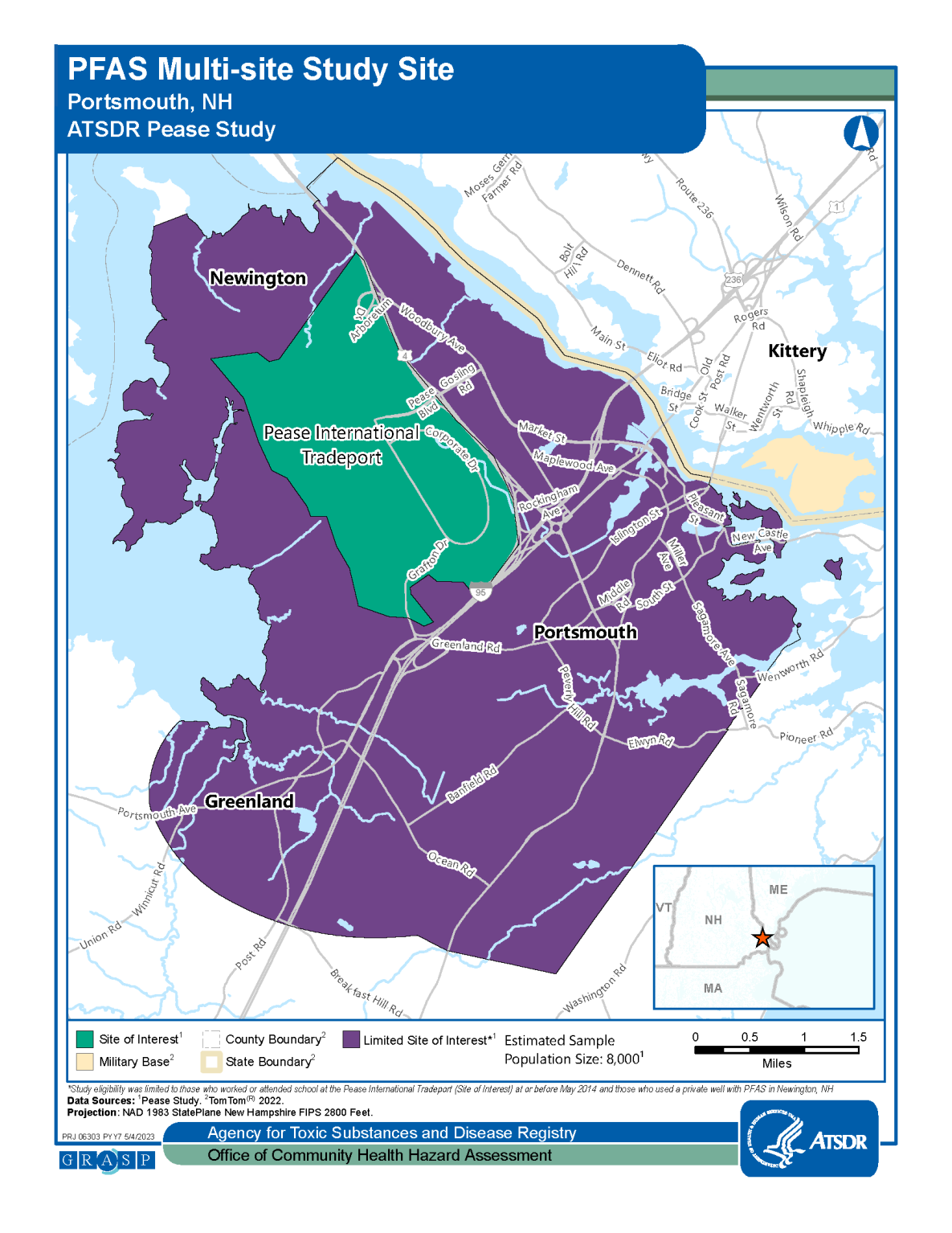
Approximately 8,000 people work at or frequent the Pease International Tradeport. There are also two daycare centers located on the site. In May 2014, drinking water wells that supply the Pease International Tradeport were sampled. The Haven Well, one of three wells that serves the Pease International Tradeport and the New Hampshire Air National Guard base at Pease, showed elevated levels of the unregulated contaminant perfluorooctane sulfonic acid (PFOS). How long PFOS may have been present in this well is not clear.
Because the level of perfluorooctane sulfonic acid (PFOS) exceeded the “provisional health advisory” set by the U.S. Environmental Protection Agency (EPA), the well was shut down by the City of Portsmouth on May 12, 2014, and since that time it has been physically disconnected from the system. The New Hampshire Department of Health and Human Services asked CDC/ATSDR to help evaluate possible follow-up actions.
Background materials:
- Pease Feasibility Assessment [PDF -1MB]
- Archived Materials
Interested in learning more about ATSDR activities at the Pease International Tradeport Site? Join us for the next CAP meeting. Click here for more information about the Pease CAP.
The next Pease CAP Meeting will be held on January 30, 2024.
The below materials were developed during the enrollment phase of the Pease Study and used for community outreach. Enrollment for the Pease Study ended December 2021. Some of the products and materials listed are provided for historical context and are no longer being updated.
Click here for Archived Materials.
For general information about PFAS or the Pease Study call, 1-800-CDC-INFO (800-232-4636).
For general questions about the Pease Study, email PeaseStudy@cdc.gov.