Part 4 – Section 11
Management Strategy 3: Medications Atropine
- Learning Objectives
- Atropine: Mechanism of Action
- Diagnostic Use: The “Atropine Challenge”
- Atropine: Available Forms (Optional Reading)
- Atropine: Stability of Solution (Optional Reading)
- Atropine: Route of Administration
- Misconception
- Reality
- Reasons Why Signs of Atropinism Are Not the Appropriate End Point to Guide Atropine Treatment
- Atropine Dose
- Misconception
- Reality
- Total Doses of Atropine in Poisoning with Organophosphorus Compounds (Optional Reading)
- Extremely High Total Doses of Atropine May be Needed in Suicidal Ingestions of Organophosphorus Compounds (Optional Reading)
- Nerve Agents Require Lower Total Doses of Atropine (Optional Reading)
- Carbamates Require Lower Doses (Optional Reading)
- Atropine: Low Incidence of Adverse Effects
- Alternatives to Atropine (Optional Reading)
- Key Points
- Progress Check
Upon completion of this section, you should be able to identify the:
- Mechanism by which atropine counters the effects of the cholinergic toxidrome.
- Clinical findings against which to titrate atropine dosage.
- Preferred routes of administration of atropine.
- Type of cholinesterase inhibitor toxicity that may require extremely high doses of atropine.
Muscarinic effects
Atropine works by competitively occupying muscarinic receptor sites, thus reducing the effects of excessive acetylcholine on these sites brought about by cholinesterase inhibition.
Nicotinic effects
Atropine is not thought to have significant effect on nicotinic receptors, and thus does not counteract fasciculations, weakness, or flaccid paralysis. (Leikin, Thomas et al. 2002) Thus, even when given sufficient doses of atropine, patients may need artificial ventilation, sometimes for weeks. (Singh, Batra et al. 1995)
Note: Although some have suggested that atropine does not cross the blood-brain barrier to any significant extent, (Finkelstein, Kushnir et al. 1989) others disagree, having noted prompt resolution of CNS symptoms after its administration. (Sofer, Tal et al. 1989; De Wilde, Vogelaers et al. 1990)
A number of authors have recommended the “atropine challenge” as an aid to diagnosis.
When given to a normal person who has not been exposed to cholinesterase inhibitors, a 2 mg dose of atropine (0.025-0.050/kg in pediatric cases) causes:
- A dry mouth.
- An increase in heart rate of about 35 beats/minute (which is usually not noticed by the recipient) within 3-5 minutes of an I.V. dose, and a maximal increase in heart rate of about 35-45 beats/minute with I.M. or autoinjector administration, respectively, within about 35-45 minutes (the longer being with I.M. injection).
- Blurred near-vision.
- Dry, hot skin.
- Mydriasis (pupillary dilation).
Most of these effects will dissipate within 4-6 hours, except blurred near-vision which may persist for 24 hours. (Sidell 1997)
It has been suggested that when these physiological changes do not occur with this dose (sometimes referred to as an atropine challenge), this is indicative of cholinesterase inhibitor toxicity. (Clark 2002; Erdman 2004)
Cautions
- If miosis (pupillary constriction) is due to direct conjunctival vapor exposure, it is relatively unresponsive to parenteral atropine. (Durham and Hayes 1962; Tareg et al. 2001) (Although, it does respond to topical administration).
- In 2-13% of cases of cholinesterase inhibitor toxicity, mydriasis (pupillary dilation) — rather than miosis (pupillary constriction), (Tsachalinas, Logaras et al. 1971; Hayes, van der Westhuizen et al. 1978; Aaron and Howland 1994; Erdman 2004)and tachycardia — rather than bradycardia (3-77% of cases), (Tsachalinas, Logaras et al. 1971; Hayes, van der Westhuizen et al. 1978; Goswamy, Chaudhuri et al. 1994; Nouira, Abroug et al. 1994; Singh, Batra et al. 1995; Clark 2002; Erdman 2004) may be a presenting signs.
- One author points out that this strategy has never been empirically tested and may not be very sensitive or specific. (Erdman 2004)
- Parenteral atropine is not generally recommended for those whose sole manifestation of toxicity is miosis (pupillary constriction). (Sidell 1997)
- Some cases of mild to moderate poisonings may improve with these doses of atropine. Thus, signs of atropinization do not always exclude the presence of cholinesterase inhibitor toxicity. (Clark 2002)
The available forms of atropine are
- Automatic injectors: the U.S. Armed Forces uses autoinjectors, containing 2 mg of atropine. (These atropine autoinjectors are packaged with a 2-PAM autoinjector, called Mark I kits.). (Leikin, Thomas et al. 2002)
- Single or multidose vials (0.4-1 mg/ml). (Leikin, Thomas et al. 2002; Fernández 2004)
- Ophthalmic ointment (1% in 1g and 3.5 g sizes). (Billups and Billups 2002)
- Ophthalmic solution (1% in 1 ml, 2 ml, 5 ml, 15 ml sizes; 2% in 2 ml size). (Billups and Billups 2002)
- Prepackaged syringes (0.05 mg/ml, 0.5 mg/5 ml, 1 mg/10 ml). (Fernández 2004)
- Reconstituted solution from bulk powder (2 mg/ml in 3 ml syringes) (Geller 1999; Kozak, Siegel et al. 2003) Discussed later in this section under the topic, Antidote Stocking.
Caution: One author suggest avoiding large doses of pre-mixed atropine containing alcohol preservatives in children out of concern that alcohol toxicity could complicate the situation. (Schenker, Louie et al. 1998)
There is evidence that atropine solution is very stable and can retain potency long after its expiration date. The table below shows several samples of solution, dating back to World War II, showing its potency. Presumably, the solution was stored in unopened containers (as opposed to multi-dose vials from which any doses had been previously extracted, although this was not explicitly stated. (Schier, Ravikumar et al. 2004)
| Expiration Date | Labeled Conc. (µgml) | Measured Conc. | 95% Confidence Intervals |
|---|---|---|---|
| Unexpired | 400 | 252 | (235-268) |
| 2001 | 400 | 290 | (272-308) |
| 1999 | 400 | 314 | (295-333) |
| 1990 | 400 | 398 | (375-420) |
| World War II | 2,000 | 1,475 | (1,385-1,565) |
Note: Tropine, a marker of degradation was not found in substantial amounts in any of the samples.
Data from: Schier JG, Ravikumar PR, Nelson LS, Heller MB, Howland MA, Hoffman RS. Preparing for chemical terrorism: Stability of injectable atropine sulfate. Academic Emergency Medicine. April 2004; 11:329-334.
In approximate order of preference, the following routes of administration can be used for the administration of atropine
- Intravenous: bolus, followed by I.V. drip. (Wiener and Hoffman 2004)
- Intraosseous: (American Heart Association 2005) bolus, followed by continuous infusion.
- Military MARK I atropine autoinjector: Although intravenous injection is the preferred route of administration, use of the autoinjector may be more practical in the field, where it can be rapidly administered even through clothing.) Blood levels are achieved more rapidly than by other forms of IM injection. Note that each MARK I kit contains an atropine autoinjector, containing 2 mg of atropine plus another autoinjector containing 600 mg of 2-PAM. (Sidell 1997; Wiener and Hoffman 2004) Pediatric atropine autoinjector syringes are now available in 0.5 mg and 1 mg sizes. (Newmark 2004) (See Figures 10-12.)
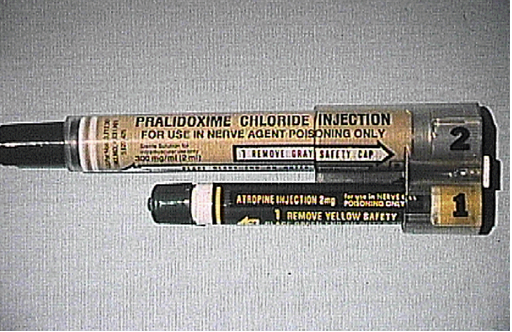
Figure 10. Military MARK I Kit containing atropine and 2-PAM autoinjectors.
Source: U.S. Army Soldier and Biological Chemical Command (SBCCOM).
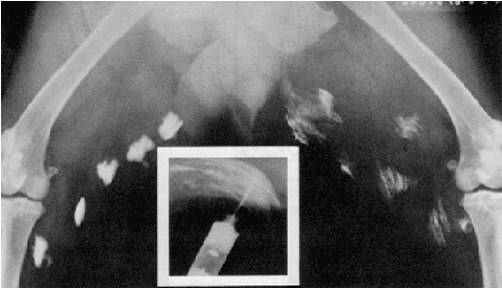
Figure 11. X-ray showing autoinjector effectiveness. The rapid absorption of antidote following automatic injection is enhanced by the degree of tissue dispersion achieved by the auto-injector. The X-ray shows autoinjector doses (on left) compared to standard syringe IM doses (on right). The autoinjector medication is obviously more efficiently diffused into surrounding muscle due to the force with which it is expelled from the injector (as seen in inset photo.) Source: U.S. Army, SBCCOM. (Note: the correct way to view an X-ray is as if the patient was facing you. Thus, the patient’s left side is shown on the right.)
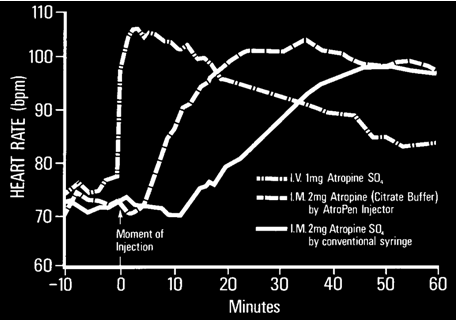
Figure 12. The effects of atropine versus route and method of administration. Source: U.S. Army Soldier and Biological Chemical Command (SBCCOM).
-
- Intramuscular: Research for this Case Study did not turn up any comparisons of intramuscular with inhalation routes of atropine administration.
- Inhalation: by nebulized inhalation (Wiener and Hoffman 2004) or via the intratracheal route. The intratracheal route can be used, but absorption is notably less complete and less reliable than the intravenous or intraosseous routes, which are preferred. The optimal intratracheal dose is unknown, but is typically administered in an amount 2-2½ times the intravenous dose. The American Heart Association recommends that the dose be diluted in 5-10 ml water or normal saline. (Reigart and Roberts 1999; American Heart Association 2005; American Heart Association 2005)
- Oral: use has been reported after I.V. administration became unnecessary. (du Toit, Muller et al. 1981)
- Ophthalmic: Anticholinergic eye drops (e.g., atropine or homatropine) have been recommended for severe eye pain caused by miosis (pupillary constriction), and secondary reflex nausea and vomiting, but may result in blurred vision. (Durham and Hayes 1962; Sidell 1997) However, one author questions whether there is enough evidence to recommend this practice. (Wiener and Hoffman 2004)
Atropine should be administered until signs of atropinism appear (tachycardia, pupil dilation, and dry mouth).
Although atropine has been used to control other, non-life-threatening effects (e.g., nausea and vomiting) the most crucial end-point for atropine dosage titration is control of clinically significant bronchorrhea, bronchoconstriction, (as reflected by level of oxygenation and ease of ventilation) and dangerous bradyarrhythmias or AV-blocks. (Sidell 1997; Reigart and Roberts 1999; Taylor 2001; Erdman 2004; Wiener and Hoffman 2004)
Tachycardia should not be used as an end-point, because it sometimes is a nicotinic manifestation of toxicity.
Resolution of miosis [Miosis has been defined as pupillary diameter of <3 mm in the dark, along with sluggish or absent response to light. (Gaon and Werne 1955)] should not be used as an end-point, because:
- Miosis (pupillary constriction) from systemic exposure may be a late finding. (Sidell 1997)
- When miosis pupillary constriction) is present, it may be resistant to systemic atropine therapy. (Durham and Hayes 1962; Sidell 1997; Tareg et al. 2001)
- Miosis (pupillary constriction) may reflect only localized ophthalmic exposure to vapor without systemic effects. (Durham and Hayes 1962; Sidell 1997)
- Pupils are of normal size in a significant minority of poisoned patients (20% in one series). (Tsachalinas, Logaras et al. 1971)
- Toxic patients may present with mydriasis (pupillary dilation) due to occasional dominance of nicotinic effects from cholinesterase inhibitors. (Clark 2002; Erdman 2004)
Adults
The most commonly recommended initial doses range from 2 to 6 mg (0.02-0.04 mg/kg). (du Toit, Muller et al. 1981)
Authors differ with regards to how frequently these doses should be titrated. Recommended dose intervals vary widely from every 2 to every 30 minutes (Willems 1981; Goswamy, Chaudhuri et al. 1994; Singh, Batra et al. 1995; Carlton, Simpson et al. 1998; Schenker, Louie et al. 1998; Tareg et al. 2001; Erdman 2004; Fernández 2004) (or 1-3 2 mg autoinjector doses). (Sidell 1997)
After adequate control of secretions, Du Toit et al. started their patients with an I.V. maintenance drip of 0.02-0.08 mg/kg/hr titrated to effect. (du Toit, Muller et al. 1981) One reported case required 0.5-2.4 mg/kg/hr, I.V. drip, during 5-weeks of treatment. (LeBlanc, Bensen et al. 1986)
Some attempts have been made (mostly for nerve agents) to characterize the doses needed according to severity of symptoms. (See the table below for an example.)
The goal of therapy with atropine is to reverse life-threatening signs and symptoms (i.e., respiratory distress), and make the patient more comfortable. Currently it is thought that this does not necessarily require the reversal of all effects of the cholinesterase inhibitor [e.g., miosis (pupillary constriction)].
Generally, in patients with severe symptoms, it is better to give too much atropine than too little. (Sidell 1997)
Children under 12 years of age
Most authors’ recommended doses range from 0.05-0.1 mg/kg boluses q 2-30 min. (Zwiener and Ginsburg 1988; Carlton, Simpson et al. 1998; Reigart and Roberts 1999; Fernández 2004)
Pediatric atropine autoinjectors (0.5 mg, 1 mg sizes) (Food and Drug Administration 2003) See the chart below.
| Patient Weight | Dose |
|---|---|
| 15-40 lbs | One 0.5 mg autoinjector dose |
| 40-90 lbs | One 1 mg autoinjector dose |
Intravenous drip
Recommendations for I.V. maintenance doses have ranged from 0.2-2.0 mg/hour (0.025 mg/kg/hr in children). (du Toit, Muller et al. 1981; Erdman 2004)
Ophthalmic
Topical mydriatics, such as atropine, and homatropine, can provide relief from eye pain and reflex nausea and vomiting. However, these drugs cause blurring of vision, and should be reserved for cases with severe eye pain. (Sidell 1997)
Warning: Hydrocarbons may be used as diluents in liquid formulations of cholinesterase inhibitors. In cases of ingestion, aspiration pneumonitis with acute respiratory distress syndrome may add to the muscarinic respiratory effects of the poison, but is unresponsive to atropine. (Reigart and Roberts 1999)
The dosage requirements for organophosphorus pesticide toxicity, especially with suicidal ingestions, can be higher by orders of magnitude than is the case for nerve agents.
Organophosphorus compounds
The range of atropine doses in the first 12-24 hours stratified by severity is illustrated in the following tables.
| Severity of Poisoning | Mean Dose (mg) +/- Standard Deviation Over First 24 hrs |
|---|---|
| Mild (mild muscarinic signs such as lacrimation, miosis (pupillary constriction), excessive sweating, and hypersalivation). | 6.0 +/- 0.7 mg |
| Moderate (partial or full-blown spectrum of symptoms, but who were breathing unassisted). | 26.1 +/- 6.5 mg |
| Severe (requiring assisted ventilation). | 49.8 +/- 4.5 mg |
Note: Guidelines used to gauge atropine dosage were documented in only 32 patients. Signs of mild atropine over dosage or dry skin and mucous membranes were used in 18 cases. Tachycardia or mydriasis (pupillary dilation) alone was used in 14 cases.
Data source: Hirshberg and Lerman, 1984. (Hirshberg and Lerman 1984)
In cases of suicidal ingestions, atropine doses in the hundreds of milligrams per day have sometimes been needed. (Wyckoff, Davies et al. 1968; Hopmann and Wanke 1975; du Toit, Muller et al. 1981; Golsousidis and Kokkas 1985; Goswamy, Chaudhuri et al. 1994; Singh, Batra et al. 1995)
- In one reported case, 3,600 mg of atropine was administered in a 24 hour period, with a total dose of 30,730 mg over the patient’s 35 days of treatment. Treatment was maintained over a 5 week period at 0.5-2.4 mg/kg/hr, I.V. drip. (LeBlanc, Bensen et al. 1986)
- A case reported by Wyckoff (1968) required a total of 1,122 mg of atropine over 10 days. (Wyckoff, Davies et al. 1968)
- Willems (1981) reported on some patients who received maintenance atropine doses for up to 20 days. (Willems 1981)
In general, severe nerve agent poisoning requires lower total doses of atropine than for organophosphorus compounds. (Sidell 1997)
In severe cases (apneic and unconscious), it may require up to 5-15 mg of atropine to restore consciousness and breathing, and atropine has not been required for more than 2-3 hours. (However, distressing, but not life-threatening effects, such as nausea and vomiting, have required atropine for 6-36 hours afterwards). (Sidell 1997)
In the Tokyo Sarin attack, only 21 of 107 patients who needed atropine required more than 2 mg, and none required more than 9 mg. (Okumura, Takasu et al. 1996)
| Severity | Signs and Symptoms | Size of titrating doses, q 5-10 min, PRN, I.V.*, or I.M. |
|---|---|---|
| Minimal | Isolated miosis (pupillary constriction) and/or rhinorrhea +/- reflex nausea and vomiting. | None‡ |
| Mild | Mild dyspnea, if it does not improve 15-30 after removal from exposure. (Localized sweating, fasciculations after dermal exposure). | 0-2 mg‡, or 0-1 MARK 1 kit |
| Moderate | Moderate to severe respiratory, G.I., or Neuromuscular findings. | 2-4 mg, or 1-2 MARK 1 kits |
| Severe | Apnea, flaccid paralysis, coma, convulsions; severe respiratory distress, or G.I. findings. | 6 mg, or 3 MARK 1 kits |
*I.V. is the preferred route, when feasible.
‡ If < 5 minutes since exposure, increase dose to next level.
Data sources: Leikin et al. (Leikin, Thomas et al. 2002) and Sidell et al. (Sidell 1997)
While it is difficult to find information on the actual doses of atropine needed, the severity of carbamate poisoning tends to be less than that for organophosphorus compounds.
The duration of toxicity also tends to be shorter for most patients; on the order of 6-12 hours. (Carlton, Simpson et al. 1998)
Warning: Mixed poisoning with organophosphorus compounds and carbamates are common. In one case series of 52 patients, 35% were mixed exposures. (Carlton, Simpson et al. 1998)
Serious, life-threatening adverse effects from the use of atropine to treat cholinesterase inhibitor toxicity appear to be uncommon. This is even when administered accidentally to children without cholinesterase inhibitor toxicity.
Example: In 268 children accidentally autoinjected with high-dose (up to 17 fold higher than recommended for age) in Israel during the Persian Gulf crises, no fatalities, seizures or life-threatening dysrhythmias were observed. (Amitai, Almog et al. 1992)
I.V. atropine has caused ventricular fibrillation in hypoxic animals with nerve agent poisoning. Therefore, it has been recommended that hypoxia be corrected if possible prior to atropine administration. However, atropine should not be withheld due to fears of this complication. (Leikin et al. 2002)
Excessive doses of atropine can cause a number of mostly minor anticholinergic symptoms (an exception, perhaps, being delirium), though they can last 24-48 hours. Examples include
- Blurred vision (Leikin, Thomas et al. 2002)
- Dry mouth (Leikin, Thomas et al. 2002)
- Inability to sweat. (Leikin, Thomas et al. 2002)
- Muscle fasciculations (Tareg et al. 2001)
- Mydriasis (pupillary dilation) (Leikin, Thomas et al. 2002)
- Paralytic ileus (du Toit, Muller et al. 1981)
- Pyrexia (Tareg et al. 2001)
- Tachycardia (Leikin, Thomas et al. 2002)
- Urinary retention (Sidell 1974; Clark 2002; Leikin, Thomas et al. 2002)
Note: Physostigmine should NOT be administered for these effects in patients with cholinesterase inhibitor poisoning. (Leikin, Thomas et al. 2002)
While other antimuscarinic agents (e.g., scopolamine) can counteract the effects of cholinesterase inhibitors, their inherent toxic effects in patients who do not have cholinesterase inhibitor poisoning have led to their rejection in favor of atropine. (Sidell 1997; Wiener and Hoffman 2004)
Glycopyrrolate in doses of 1-2 mg, I.V., (0.025 mg/kg in children) has been suggested as an alternative to atropine, and is said to have fewer CNS side effects. (Clark 2002) However, its use has not been extensively evaluated. (Erdman 2004)
- Atropine competitively inhibits the effects of cholinesterase inhibitors at muscarinic, but not nicotinic cholinergic receptors.
- The failure to develop signs of atropinization after a 2 mg dose (0.025-0.050 in pediatric cases) is thought to be very suggestive of cholinesterase inhibitor poisoning, although this has not been empirically tested.
- Atropine should be titrated to achieve control of clinically significant bronchorrhea and bronchoconstriction, (as reflected by level of oxygenation and ease of ventilation) and to treat dangerous bradyarrhythmias or AV-blocks.
- Pupil size and heart rate are not reliable end-points for titration of atropine.
- Extremely large doses of atropine may be necessary for poisoning due to suicidal ingestions of organophosphorus pesticides.
- In order of preference, the best routes for administration of atropine are: intravenous, autoinjector, and intramuscular.