Public Health Assessment Guidance Manual (PHAGM)

The PHAGM outlines methods and resources that environmental health professionals can use to evaluate environmental exposures associated with an industrial facility, commercial facility, or hazardous waste site.
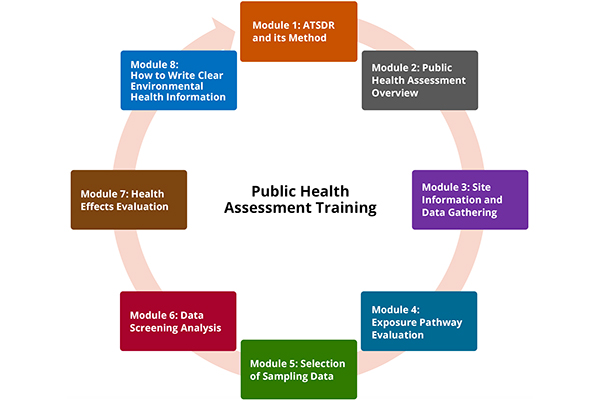
ATSDR's Public Health Assessment Training (PHAT) modules help environmental health professionals understand the public health assessment process.


[ATSDR] Agency for Toxic Substances and Disease Registry. 2022. Public health assessment guidance manual. Atlanta: US Department of Health and Human Services [accessed {insert PHAGM access date}]. Available from: https://www.atsdr.cdc.gov/pha-guidance/index.html.